Our Services:
Patients & Volunteers
Our Services:
Patients & Volunteers
A Study Participant’s Journey
With a patient-centered approach, CIC Neurosciences makes it a point of honor to simplify every step of the process. We guarantee a clear path, efficient and secure through all stages of our clinical trials. Here is an overview of our process in a few simple steps:
Preliminary visit
A patient referred by their neurologist or a healthy volunteer enrolled in the CIC registry
Eligibility
To ensure the safety of participants and the consistency of the study data, the criteria are primarily medical
Information Sheet and Consent Forms
Provides information on how to participate in the study (objectives, duration, examinations, benefits and risks, schedule of visits)
Time for reflection
You have been given some time to think it over so that you can confirm your participation if you wish.
Inclusion visit
Meeting with the research team to explain the study process again. Signing the consent form.
Follow-up visit
During routine visits, you perform various assessments: clinical and neurological examinations, gait tests, MRIs, etc.
End of study
The fact that your participation has ended does not mean that the study as a whole is over. There may be a long delay between the end of your participation and the release of the study results.
A Study Participant’s Journey
With a patient-centered approach, CIC Neurosciences makes it a point of honor to simplify every step of the process. We guarantee a clear path, efficient and secure through all stages of our clinical trials. Here is an overview of our process in a few simple steps:
Preliminary visit
A patient referred by their neurologist or a healthy volunteer enrolled in the CIC registry
Eligibility
To ensure the safety of participants and the consistency of the study data, the criteria are primarily medical
Information Sheet and Consent Forms
Provides information on how to participate in the study (objectives, duration, examinations, benefits and risks, schedule of visits)
Time for reflection
You have been given some time to think it over so that you can confirm your participation if you wish.
Inclusion visit
Meeting with the research team to explain the study process again. Signing the consent form.
Follow-up visit
During routine visits, you perform various assessments: clinical and neurological examinations, gait tests, MRIs, etc.
End of study
The fact that your participation has ended does not mean that the study as a whole is over. There may be a long delay between the end of your participation and the release of the study results.
The main conditions
The CIC Neurosciences focuses on a wide range of diseases and conditions affecting the nervous system in patients aged 15 years and 3 months or older.
Abnormal Movements
Abnormal movements result from dysfunction of the basal ganglia circuit, involving degenerative disorders (Parkinson's disease, etc.) or functional disorders (dystonia, tremors).
The CIC Neurosciences explores these disorders through multimodal studies to better understand their mechanisms and develop innovative diagnostic and therapeutic tools aimed at alleviating symptoms and slowing the progression of these diseases.
Inflammatory diseases of the central nervous system
Research on inflammatory diseases of the central nervous system (multiple sclerosis, optic neuromyelitis) focuses on three main areas: understanding the underlying mechanisms through advanced imaging (MRI, PET, retinal analysis), developing innovative therapies to better control neuroinflammation and remyelination, and optimizing current care by adapting treatments to minimize side effects.
Amyotrophic Lateral Sclerosis (ALS) or Charcot's disease Charcot's disease
Research on amyotrophic lateral sclerosis (ALS) aims to better understand the mechanisms of the disease through the analysis of clinical, biological, and genetic data, and to develop new treatments through clinical trials targeting neurodegeneration, motor function, and genetic mutations (such as SOD1).
These advances already make it possible to use certain innovative treatments under compassionate use.
Rare neurogenetic disorders
Research into rare neurogenetic diseases, such as spinocerebellar ataxias and Huntington’s disease, aims to understand the underlying biological mechanisms, identify biomarkers, and develop treatments, particularly through gene therapy.
International clinical trials are testing innovative approaches, such as reducing abnormal proteins (huntingtin) or therapies targeting the presymptomatic stages, to optimize treatments.
The CIC Neurosciences also investigates other neurological conditions, such as epilepsy, stroke, Alzheimer’s disease, frontotemporal dementia, psychiatric disorders (such as depression and obsessive-compulsive disorder), and leukodystrophies.
The main conditions
The CIC Neurosciences focuses on a wide range of diseases and conditions affecting the nervous system in patients aged 15 years and 3 months or older.
Abnormal Movements
Abnormal movements result from dysfunction of the basal ganglia circuit, involving degenerative disorders (Parkinson's disease, etc.) or functional disorders (dystonia, tremors).
The CIC Neurosciences explores these disorders through multimodal studies to better understand their mechanisms and develop innovative diagnostic and therapeutic tools aimed at alleviating symptoms and slowing the progression of these diseases.
Inflammatory diseases of the central nervous system
Research on inflammatory diseases of the central nervous system (multiple sclerosis, optic neuromyelitis) focuses on three main areas: understanding the underlying mechanisms through advanced imaging (MRI, PET, retinal analysis), developing innovative therapies to better control neuroinflammation and remyelination, and optimizing current care by adapting treatments to minimize side effects.
Amyotrophic Lateral Sclerosis (ALS) or Charcot's disease Charcot's disease
Research on amyotrophic lateral sclerosis (ALS) aims to better understand the mechanisms of the disease through the analysis of clinical, biological, and genetic data, and to develop new treatments through clinical trials targeting neurodegeneration, motor function, and genetic mutations (such as SOD1).
These advances already make it possible to use certain innovative treatments under compassionate use.
Rare neurogenetic disorders
Research into rare neurogenetic diseases, such as spinocerebellar ataxias and Huntington’s disease, aims to understand the underlying biological mechanisms, identify biomarkers, and develop treatments, particularly through gene therapy.
International clinical trials are testing innovative approaches, such as reducing abnormal proteins (huntingtin) or therapies targeting the presymptomatic stages, to optimize treatments.
The CIC Neurosciences also investigates other neurological conditions, such as epilepsy, stroke, Alzheimer’s disease, frontotemporal dementia, psychiatric disorders (such as depression and obsessive-compulsive disorder), and leukodystrophies.
Practical Information
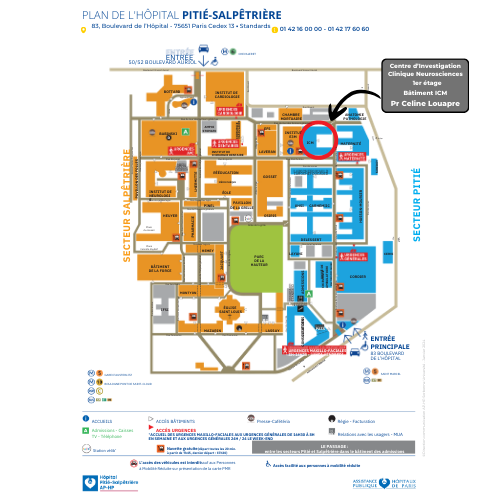
Map
Welcome Brochure

Documents to bring to the CIC:
- Your ID Card
- Your Carte Vitale is valid
- Your bank details
In case of an emergency
I am a participant in a clinical research study and I have an emergency to report or new medical information to share; I will call the phone number listed on my patient card to report these changes.
Data Management
You are participating in a clinical research study at the CIC Neurosciences: the data collected about you as part of this study will be used to achieve the study’s objectives.
When you enrolled in the study, you were given a detailed information sheet about the study and how your data will be used. Please feel free to refer to it; it covers all of the following points:
Processing of Your Data
- As part of the study, your personal data is processed to enable the analysis of the research results
- The research sponsor is responsible for the processing of your data
- The personal data collected during your visits is shared with the principal investigator or with individuals acting on behalf of the sponsor
- The data processing procedures, both during and after your participation, ensure strict confidentiality in accordance with applicable regulations
Reuse of Your Data
- Primarily in cohort studies*, the data collected about you may be reused for related analyses, with a purpose consistent with the research in which you are participating
- There are “transparency portals” that allow participants in clinical trials to stay informed about projects and analyses conducted using research or clinical care data. You will always be notified of the availability of such information channels (through the study information form, a specific letter, etc.).
Contact Information and Exercising Your Rights
For each study, there is a procedure for contacting us, obtaining information, and objecting, which allows you, if necessary, to:
- To inform you about the use and reuse of your data
- Exercise your right to withdraw from the study without affecting your medical care
- Object to the reuse of your data, if applicable
- Request access to your personal data and correct it if necessary
Feel free to discuss how your data is used with the team supporting you at the CIC!
Practical Information
Map

Welcome Brochure

Documents to bring to the CIC:
- Your ID Card
- Your Carte Vitale is valid
- Your bank details
In case of an emergency
I am a participant in a clinical research study and I have an emergency to report or new medical information to share; I will call the phone number listed on my patient card to report these changes.
Data Management
You are participating in a clinical research study at the CIC Neurosciences: the data collected about you as part of this study will be used to achieve the study’s objectives.
When you enrolled in the study, you were given a detailed information sheet about the study and how your data will be used. Please feel free to refer to it; it covers all of the following points:
Processing of Your Data
- As part of the study, your personal data is processed to enable the analysis of the research results
- The research sponsor is responsible for the processing of your data
- The personal data collected during your visits is shared with the principal investigator or with individuals acting on behalf of the sponsor
- The data processing procedures, both during and after your participation, ensure strict confidentiality in accordance with applicable regulations
Reuse of Your Data
- Primarily in cohort studies*, the data collected about you may be reused for related analyses, with a purpose consistent with the research in which you are participating
- There are “transparency portals” that allow participants in clinical trials to stay informed about projects and analyses conducted using research or clinical care data. You will always be notified of the availability of such information channels (through the study information form, a specific letter, etc.).
Contact Information and Exercising Your Rights
For each study, there is a procedure for contacting us, obtaining information, and objecting, which allows you, if necessary, to:
- To inform you about the use and reuse of your data
- Exercise your right to withdraw from the study without affecting your medical care
- Object to the reuse of your data, if applicable
- Request access to your personal data and correct it if necessary
Feel free to discuss how your data is used with the team supporting you at the CIC!
To support all of our services, CIC Neurosciences relies on state-of-the-art infrastructure and high-tech equipment. Located within the Brain Institute, our center offers exclusive access to the most advanced technical facilities and tools in the field.
To support all of our services, CIC Neurosciences relies on state-of-the-art infrastructure and high-tech equipment. Located within the Brain Institute, our center offers exclusive access to the most advanced technical facilities and tools in the field.
