Our Mission:
What is clinical research?
Our Mission:
What is clinical research?
Clinical research refers to all research conducted on humans. It focuses primarily onimproving our understanding of diseases, developing new treatments or medical devices, and diagnostic methods to ensure better patient care.
It is highly regulated, follows a specific research protocol, and is conducted only under certain conditions: obtaining the consent of the research participants, securing the necessary regulatory approvals and completing all required legal and ethical procedures, being conducted by qualified personnel, taking all necessary measures to protect volunteers, and having the objective of advancing medical knowledge.
There are two main types of clinical research:

Pathophysiological studies
They help improve our understanding of the disease and its progression over time (e.g., cohort studies, epidemiology).

Therapeutic studies
They provide scientific evidence of the efficacy and safety of a new drug, a new medical device, or a new treatment approach for a particular disease.
This is the necessary step for a new molecule to become a drug or for a new medical device to be marketed
In the process of developing new drugs, clinical trials are conducted in several phases:

TOLERANCE ASSESSMENT
First administration of the treatment in humans.
Conducted on small groups of healthy volunteers or patients to determine the recommended dose for the administration of this new treatment

EFFECTIVENESS EVALUATION
Phase II trials allow for the evaluation of not only tolerability but also the efficacy of the new treatment.
These trials are conducted on larger groups of participantswith the disease being treated. Multiple doses may be evaluated, most often in a blinded or double-blinded manner.

DEMONSTRATING THERAPEUTIC EFFICACY
Once the efficacy of a new treatment has been demonstrated, phase III comparative clinical trials evaluate its clinical benefit compared to a standard treatment or a placebo.
These trials, conducted as single-blind or double-blind studies, ensure the objectivity of the data collected. If the results are positive, a Marketing Authorization (MA) is sought, allowing the product to be marketed.

LARGE-SCALE EVALUATION
Once a drug is on the market, it is subject to a monitoring process known as pharmacovigilance to detect rare adverse effects and assess its tolerability in real-world conditions.
Phase IV trials also help refine the drug’s use in specific populations (the elderly, minors, etc.). It takes about 12 years, starting from preclinical studies, to ensure the efficacy and safety of a new treatment.
Clinical research refers to all research conducted on humans. It focuses primarily onimproving our understanding of diseases, developing new treatments or medical devices, and diagnostic methods to ensure better patient care.
It is highly regulated, follows a specific research protocol, and is conducted only under certain conditions: obtaining the consent of the research participants, securing the necessary regulatory approvals and completing all required legal and ethical procedures, being conducted by qualified personnel, taking all necessary measures to protect volunteers, and having the objective of advancing medical knowledge.
There are two main types of clinical research:

Pathophysiological studies
They help improve our understanding of the disease and its progression over time (e.g., cohort studies, epidemiology).

Therapeutic studies
They provide scientific evidence of the efficacy and safety of a new drug, a new medical device, or a new treatment approach for a particular disease.
This is the necessary step for a new molecule to become a drug or for a new medical device to be marketed
In the process of developing new drugs, clinical trials are conducted in several phases:

TOLERANCE ASSESSMENT
First administration of the treatment in humans.
Conducted on small groups of healthy volunteers or patients to determine the recommended dose for the administration of this new treatment

EFFECTIVENESS EVALUATION
Phase II trials allow for the evaluation of not only tolerability but also the efficacy of the new treatment.
These trials are conducted on larger groups of participantswith the disease being treated. Multiple doses may be evaluated, most often in a blinded or double-blinded manner.

DEMONSTRATING THERAPEUTIC EFFICACY
Once the efficacy of a new treatment has been demonstrated, phase III comparative clinical trials evaluate its clinical benefit compared to a standard treatment or a placebo.
These trials, conducted as single-blind or double-blind studies, ensure the objectivity of the data collected. If the results are positive, a Marketing Authorization (MA) is sought, allowing the product to be marketed.

LARGE-SCALE EVALUATION
Once a drug is on the market, it is subject to a monitoring process known as pharmacovigilance to detect rare adverse effects and assess its tolerability in real-world conditions.
Phase IV trials also help refine the drug’s use in specific populations (the elderly, minors, etc.). It takes about 12 years, starting from preclinical studies, to ensure the efficacy and safety of a new treatment.
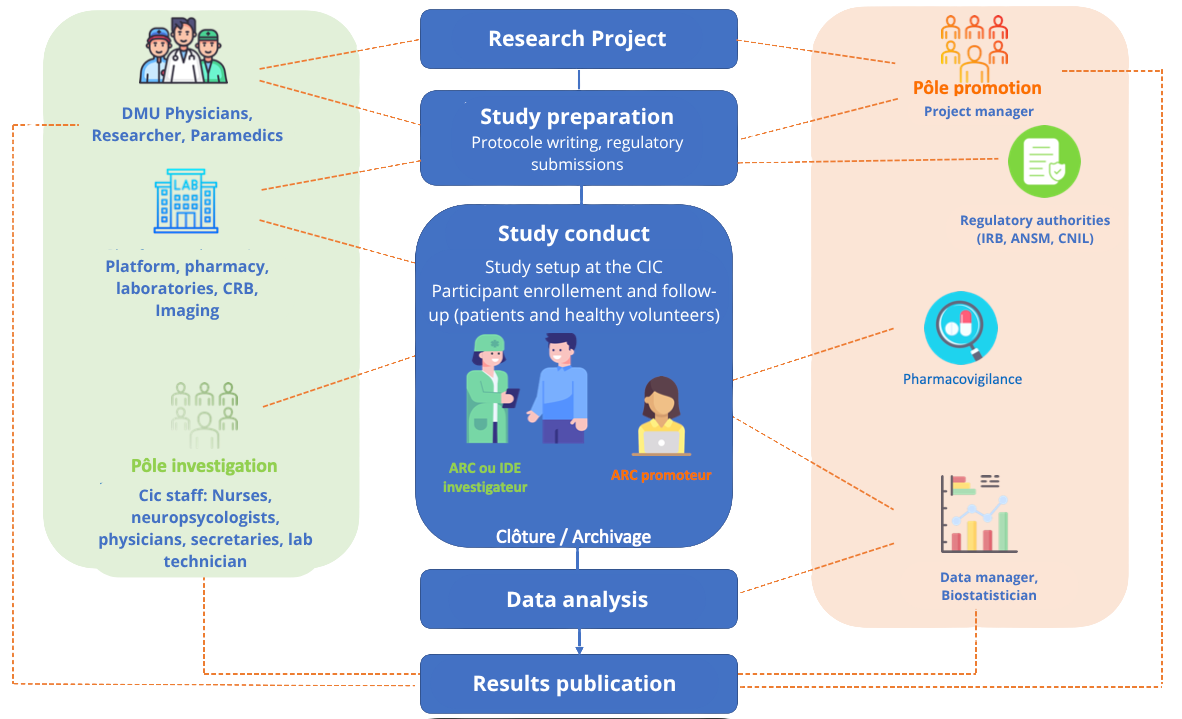
When a doctor, researcher, or healthcare professional has a research project, they begin by preparing the project by drafting the protocol, and then submit it to the regulatory authorities.
Once regulatory approvals have been obtained, the clinical trial can begin, and participants are seen at the CIC for their enrollment and follow-up visits. Following the final visit of the last participant, the trial is closed, and the participants’ medical records are archived. The data is then analyzed to meet the study’s objectives, and the results are shared through scientific publications.
Various stakeholders are involved in this process. They are part of either the investigation team (physicians, researchers, paramedical staff, support teams, and CIC staff) or the promotion team (project manager, regulatory authorities, pharmacovigilance specialists, data managers, and biostatisticians), both of which are essential to the successful completion of a research project.
The stages of a clinical trial
In order to conduct a clinical trial, it is necessary to secure one or more sources of funding.
What are the sources of funding for clinical trials?
– The government
– Charities dedicated to research
– Foundations
– Medical institutions
– Volunteer groups such as patient organizations
– The pharmaceutical industry
When a doctor, researcher, or healthcare professional has a research project, they begin by preparing the project by drafting the protocol, and then submit it to the regulatory authorities.
Once regulatory approvals have been obtained, the clinical trial can begin, and participants are seen at the CIC for their enrollment and follow-up visits. Following the final visit of the last participant, the trial is closed, and the participants’ medical records are archived. The data is then analyzed to meet the study’s objectives, and the results are shared through scientific publications.
Various stakeholders are involved in this process. They are part of either the investigation team (physicians, researchers, paramedical staff, support teams, and CIC staff) or the promotion team (project manager, regulatory authorities, pharmacovigilance specialists, data managers, and biostatisticians), both of which are essential to the successful completion of a research project.
The stages of a clinical trial

In order to conduct a clinical trial, it is necessary to secure one or more sources of funding.
What are the sources of funding for clinical trials?
– The government
– Charities dedicated to research
– Foundations
– Medical institutions
– Volunteer groups such as patient organizations
– The pharmaceutical industry
In conclusion, conducting a clinical trial requires solid expertise and close support.
Thanks to the experience and expertise of CIC Neurosciences, you benefit from a dedicated organization with extensive experience in managing research projects and hosting participants.
Our team is here to support you every step of the way, from the design phase through to post-test follow-up.
To learn more about our services or get started on your project, please feel free to contact us.
In conclusion, conducting a clinical trial requires solid expertise and close support.
Thanks to the experience and expertise of CIC Neurosciences, you benefit from a dedicated organization with extensive experience in managing research projects and hosting participants.
Our team is here to support you every step of the way, from the design phase through to post-test follow-up.
To learn more about our services or get started on your project, please feel free to contact us.
